Solids in Solution and Colligative Properties: Interactive Simulation
This simulation was prepared using Mathematica. Download the free Wolfram player, and then download the simulation CDF file (link given below or click on figure to download). Try to predict the behavior when a parameter changes before using a slider to change that parameter. A screencast below explains how to use this simulation.
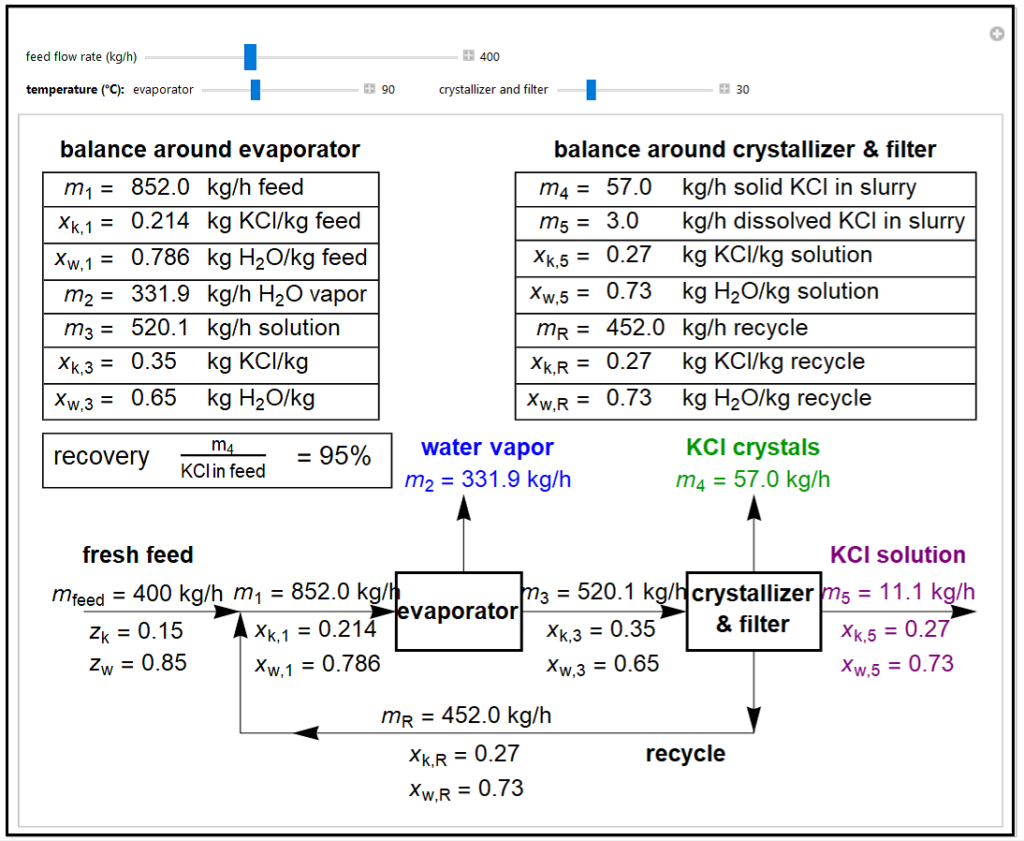
This demonstration shows mass balances for an evaporative crystallization process. A 15% potassium chloride/85% water feed is mixed with a recycle stream and fed to an evaporator, which evaporates water and increases the KCl concentration in the exiting solution. This solution enters a crystallizer and filter. A filter cake of KCl crystals and a portion of the saturated KCl solution are removed from the crystallizer and filter. The rest of the KCl solution is recycled and mixed with the fresh feed. Use sliders to set the fraction of crystals in the product stream, and the temperatures of the evaporator and crystallizer/filter. The solubility of KCl increases linearly with temperature.
Try to answer these questions before determining the answer with the simulation. We suggest that you write down the reasons for your answers.
- What happens when you increase the feed flow rate?
- How does increasing the temperature of the evaporator affect the amount of KCl solution leaving the crystallizer?