Solids in Solution and Colligative Properties: Example Problems
Try to solve these problems before watching the solutions in the screencasts.
Example Problem 1
A cooling crystallizer is used to form KNO3 crystals from a solution of the dissolved salt in water at 100°C. When 100 kg of solution is fed to the crystallizer, which operates at 30°C, 25 kg of solid KNO3 is extracted. By how many degrees C is the inlet stream superheated?
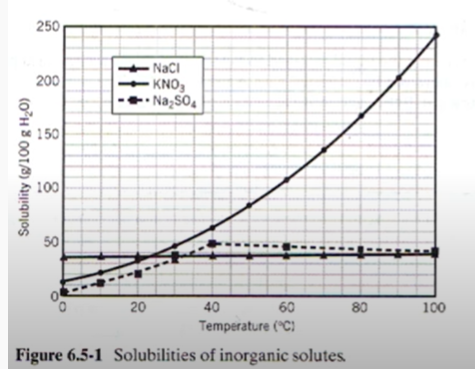
Example Problem 2
If 304.7 g of glucose is added to 1.0 L of water, what is the freezing temperature of this mixture?
Example Problem 3 (optional)
We have a crystallizer unit that is used to remove K2Cr2O7 from solution. A saturated solution of K2Cr2O7 at 60°C enters the crystallizer where the temperature is maintained at 20°C. This drop in temperature induces crystals that become entrained in the solution. The goal is to produce 200 kg of crystal solids that are filtered out of a solution that is twice the flow rate of the solids. How much water needs to be evaporated from the crystallizer to make this happen?
Using Solubility Diagrams for Material Balances



