Flash Distillation of a Benzene, Toluene, p-Xylene Mixture

Description
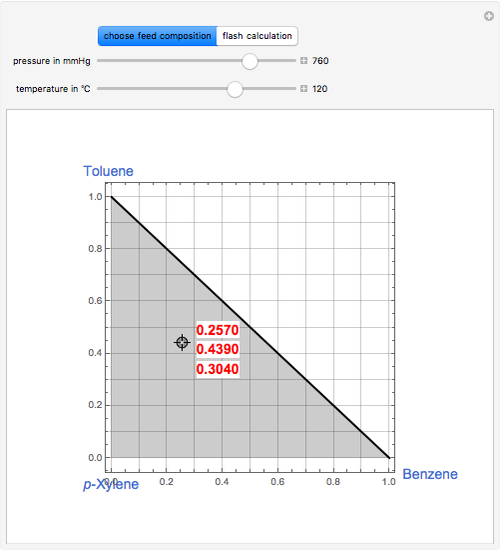
For an ideal ternary mixture of benzene, toluene, and p-xylene (BTX), vapor-liquid equilibrium data can be computed using Raoult’s law, yiP = xiPisat, where Pisat is the vapor pressure of component i given by Antoine’s equation, P is the total pressure, and xi and yi are the liquid and vapor phase mole fraction of component i. Select a feed composition by dragging the locator in the ternary triangular diagram (i.e. inside the gray region). Vary the values of P and T using the sliders. This simulation computes the vapor fraction φ of the feed and the composition of the liquid and vapor streams leaving the isothermal flash drum. Only when Tbubble < T < Tdew will the feed be a two-phase mixture such that 0 < φ < 1.
About
Authors: Housam Binous, Mamdouh Al-Harthi, Brian G. Higgins. Open content licensed under CC BY-NC-SA.
View the source code for this simulation



