Gibbs Phase Rule for One- and Two-Component Systems

Description
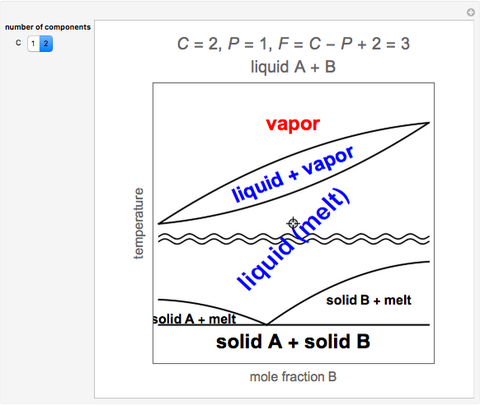
A simple rule determines the number of degrees of freedom for a heterogeneous system. If a system in thermodynamic equilibrium contains P phases and C components, then the number of degrees of freedom is given by F = C – P + 2. Degrees of freedom, F, represents the number of intensive variables (such as pressure, temperature, and composition) that can be varied arbitrarily over some finite range without changing the number of phases.
This simulation runs on desktop using the free Wolfram Player. Download the Wolfram Player here.
About
Author: S. M. Blinder. Open content licensed under CC BY-NC-SA
View the source code for this simulation



